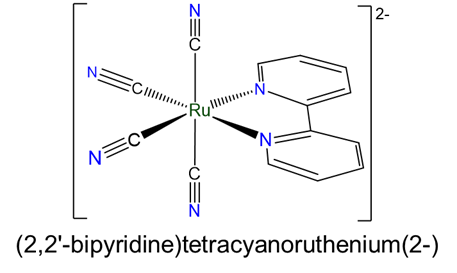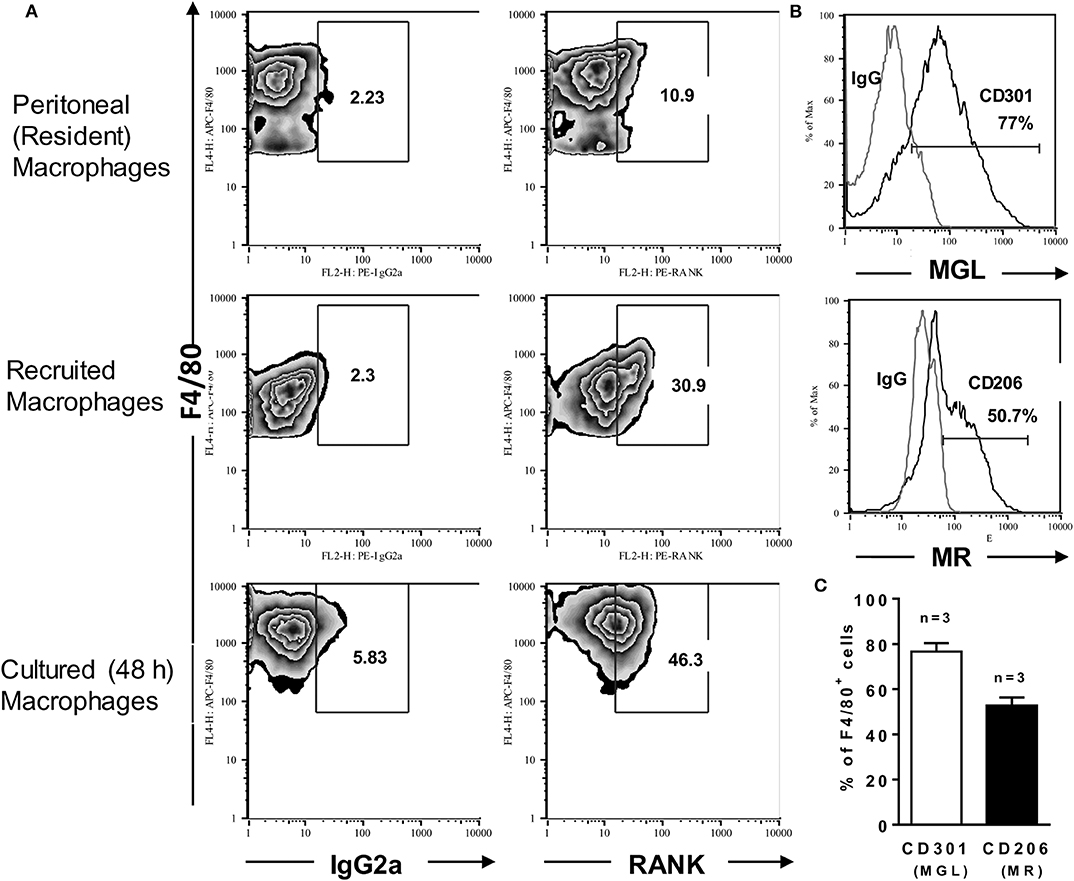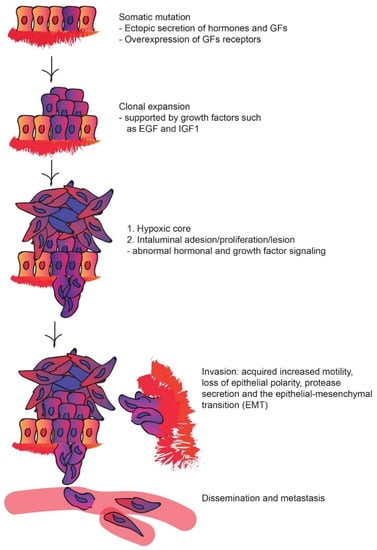
Cancers | Free Full-Text | Multifaceted Interplay between Hormones, Growth Factors and Hypoxia in the Tumor Microenvironment

Mutation of any protein binding site in the Ig 3-enhancer core region... | Download Scientific Diagram
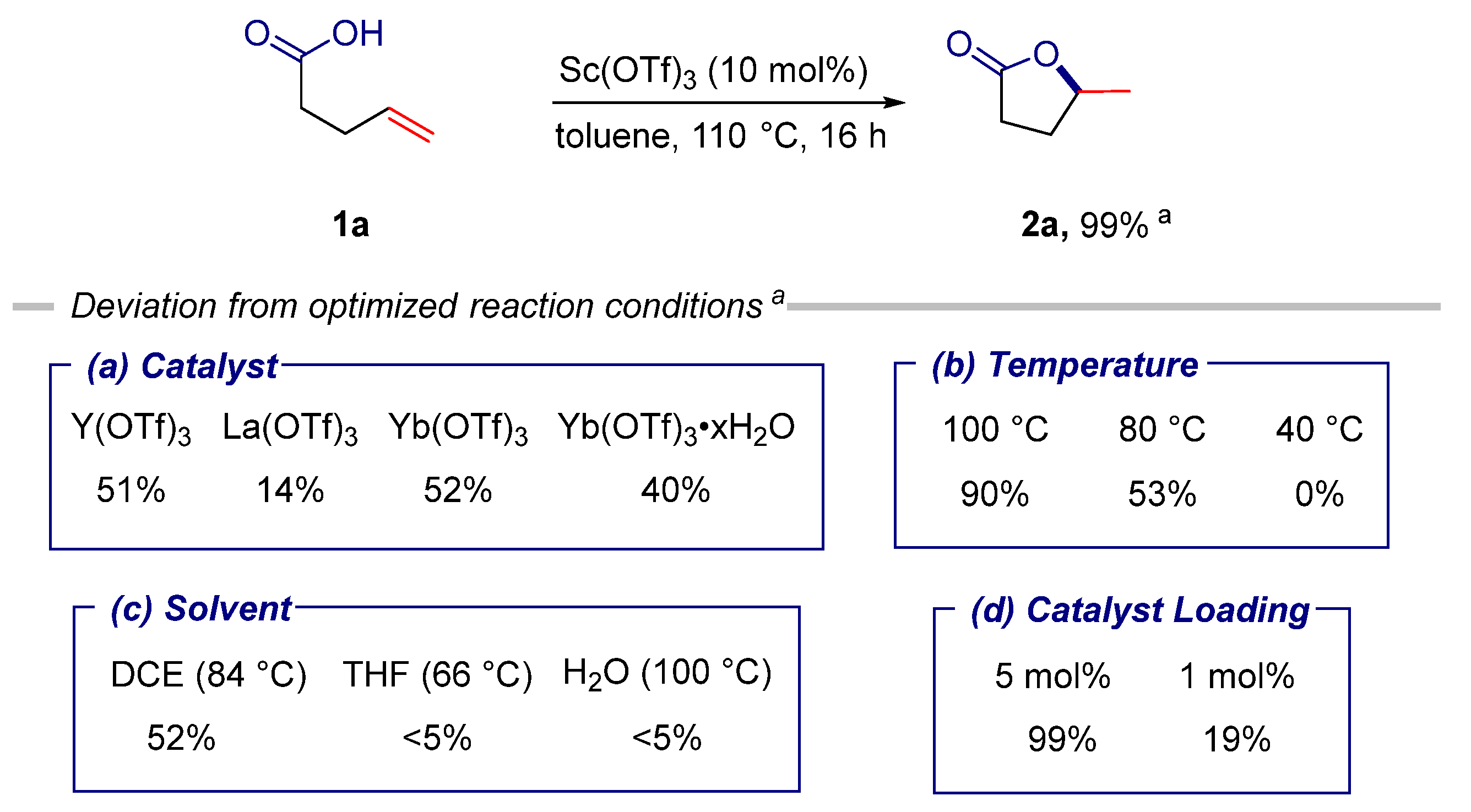
Catalysts | Free Full-Text | Substrate-Dependent Selectivity in Sc(OTf)3-Catalyzed Cyclization of Alkenoic Acids and N-Protected Alkenamides
![Reactivity of [PtCl(η2-C2H4)(N-N)]+, N-N = diimine ligand, with phenol derivatives and first comparison between single crystal X-ray structures of syn- and anti-[Pt(N-N)(phenolate)2] rotamers in the solid state - ScienceDirect Reactivity of [PtCl(η2-C2H4)(N-N)]+, N-N = diimine ligand, with phenol derivatives and first comparison between single crystal X-ray structures of syn- and anti-[Pt(N-N)(phenolate)2] rotamers in the solid state - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0020169313004945-fx1.jpg)
Reactivity of [PtCl(η2-C2H4)(N-N)]+, N-N = diimine ligand, with phenol derivatives and first comparison between single crystal X-ray structures of syn- and anti-[Pt(N-N)(phenolate)2] rotamers in the solid state - ScienceDirect
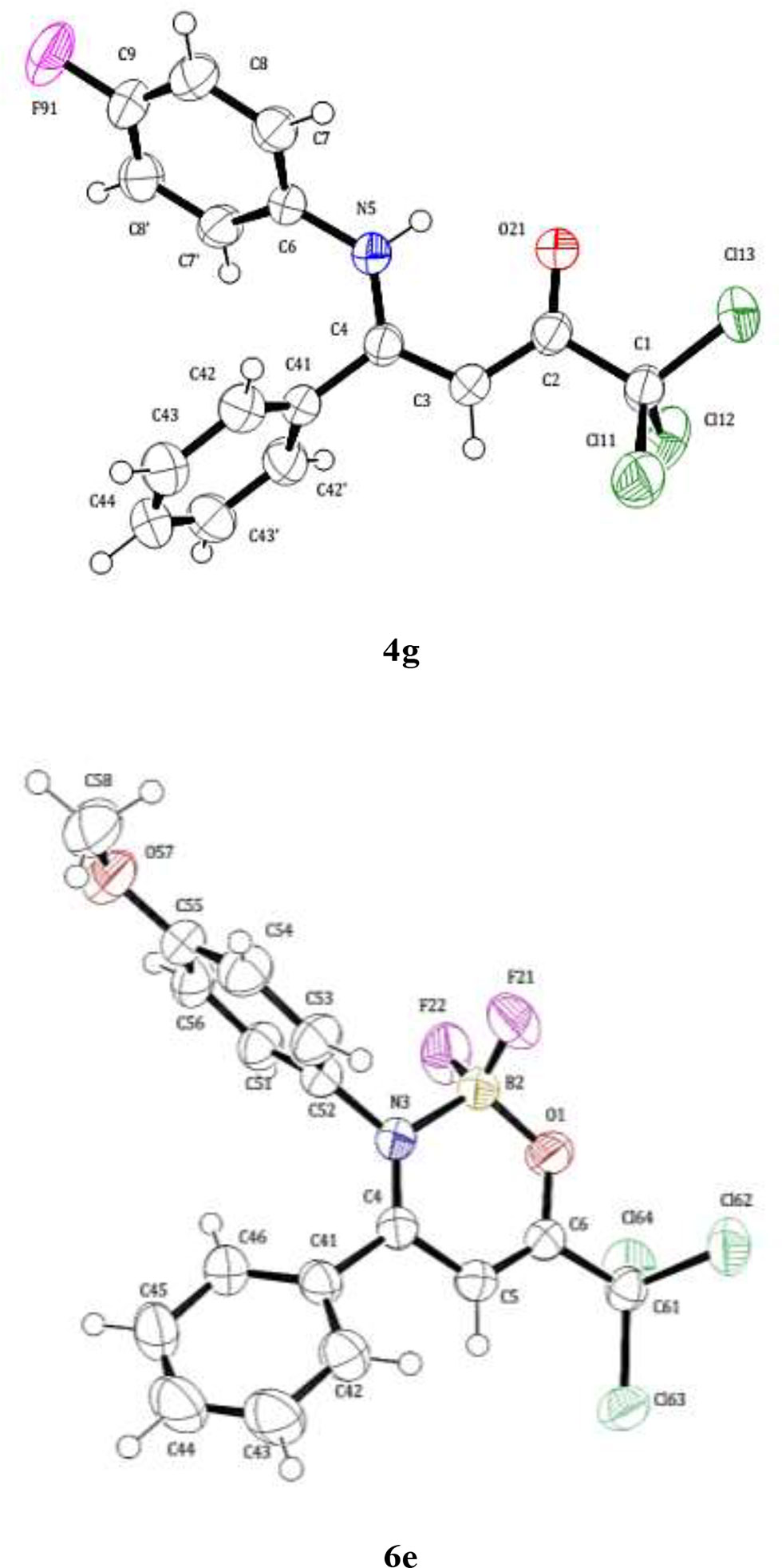
Frontiers | Novel Alkyl(aryl)-Substituted 2,2-Difluoro-6-(trichloromethyl)-2H-1,3,2-oxazaborinin-3-ium-2-uides: Synthesis, Antimicrobial Activity, and CT-DNA Binding Evaluations
![Reactivity of [PtCl(η2-C2H4)(N-N)]+, N-N = diimine ligand, with phenol derivatives and first comparison between single crystal X-ray structures of syn- and anti-[Pt(N-N)(phenolate)2] rotamers in the solid state - ScienceDirect Reactivity of [PtCl(η2-C2H4)(N-N)]+, N-N = diimine ligand, with phenol derivatives and first comparison between single crystal X-ray structures of syn- and anti-[Pt(N-N)(phenolate)2] rotamers in the solid state - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0020169313004945-gr1.jpg)
Reactivity of [PtCl(η2-C2H4)(N-N)]+, N-N = diimine ligand, with phenol derivatives and first comparison between single crystal X-ray structures of syn- and anti-[Pt(N-N)(phenolate)2] rotamers in the solid state - ScienceDirect

Lanthanide(III) Complexes with Ligands Derived from a Cyclen Framework Containing Pyridinecarboxylate Pendants. The Effect of Steric Hindrance on the Hydration Number | Inorganic Chemistry
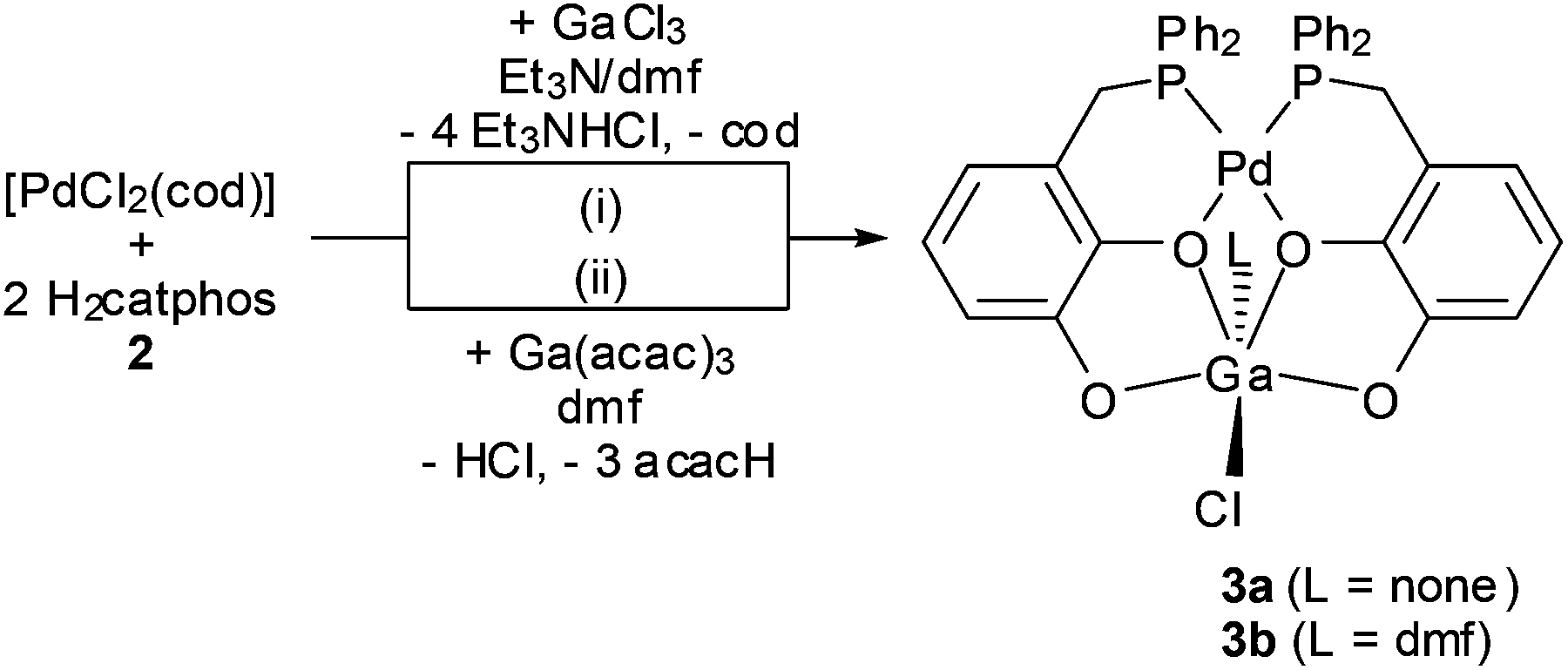
Heterobimetallic catechol-phosphine complexes with palladium and a group-13 element: structural flexibility and dynamics - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT00785A

Electronic and Structural Variations of a Nickel(0) N-Heterocyclic Phosphenium Complex in Comparison to Group 10 Analogues | Inorganic Chemistry

PDF) Six-coordinate high-spin iron(II) complexes with bidentate PN ligands based on 2-aminopyridine - new Fe(II) spin crossover systems
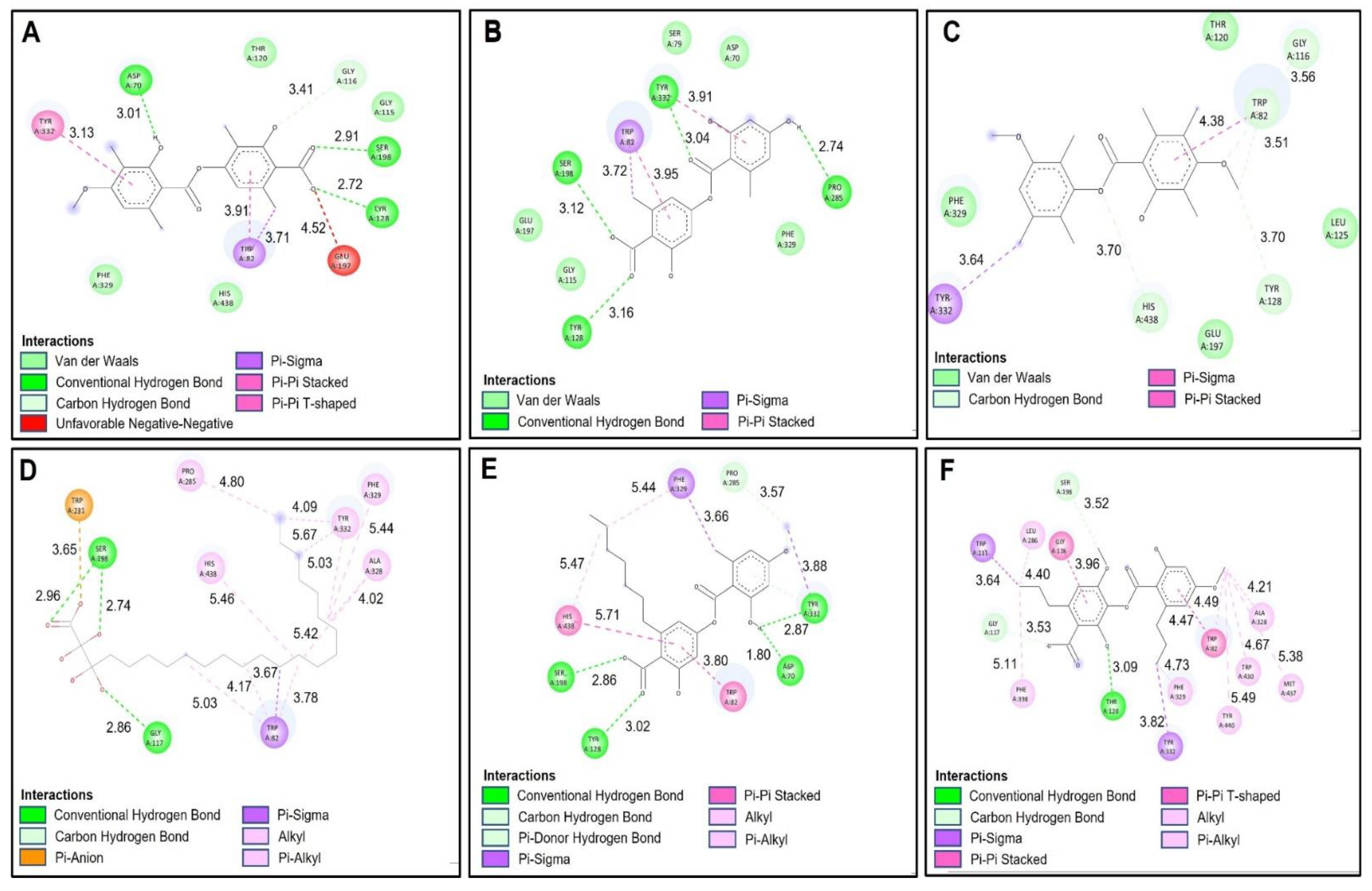
Molecules | Free Full-Text | Metabolomic Profiling, Antioxidant and Enzyme Inhibition Properties and Molecular Docking Analysis of Antarctic Lichens

Copper(I)–USY as a Ligand-Free and Recyclable Catalyst for Ullmann-Type O-, N-, S-, and C-Arylation Reactions: Scope and Application to Total Synthesis | The Journal of Organic Chemistry

PDF) Six-coordinate high-spin iron(II) complexes with bidentate PN ligands based on 2-aminopyridine - new Fe(II) spin crossover systems
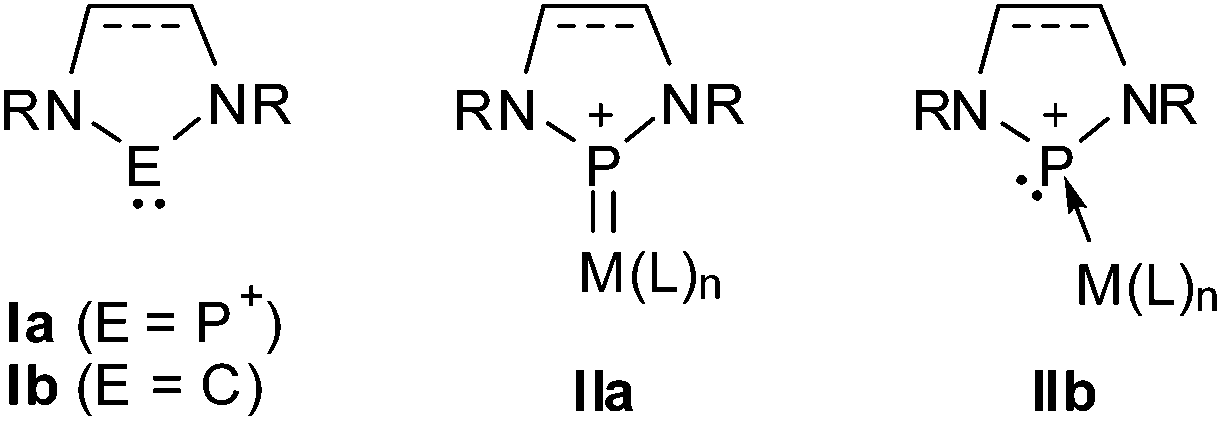
An anionic phosphenium complex as an ambident nucleophile - Dalton Transactions (RSC Publishing) DOI:10.1039/C5DT00008D

PDF) Six-coordinate high-spin iron(II) complexes with bidentate PN ligands based on 2-aminopyridine - new Fe(II) spin crossover systems